Image outlining the basics of PoreDesigner's workflow for altering the pore size of a bacterial channel protein.
PoreDesigner process improves water treatment, bioseparations
2/6/2019
UNIVERSITY PARK, Pa. — A team of researchers from Penn State and the University of Illinois at Urbana-Champaign (UIUC) have created PoreDesigner, a fully automated computational workflow process for altering the pore size of a bacterial channel protein. This enables assembly of the proteins into artificial membranes for precise sub-nanometer scale separation of solutes of marginal size difference, which can improve water purification and laboratory bioseparations.
The team’s research was a subject of a paper in the journal Nature Communications.
Natural membranes form an ideal model for synthetic membranes, as all their pores are exactly the same pore size, leading to high productivity combined with high selectivity (i.e., only molecules of the desired size and shape get through). The natural membranes have various protein channels that transport water, ions and small molecules between cells. They also block out impurities that cannot fit through the pores.
“It is difficult to introduce same pore sizes in a uniform distribution in artificial membranes, such as polymeric membranes used in industry, but doing so will allow water to go through faster and simultaneously reject solutes larger than water or another size set for solutes,” Ratul Chowdhury, doctoral candidate in chemical engineering, said. “Also, the natural pores we reengineered had a size greater than one nanometer, so we had to engineer them to access sub-nanometer sizes, which are more useful for molecular separations.”
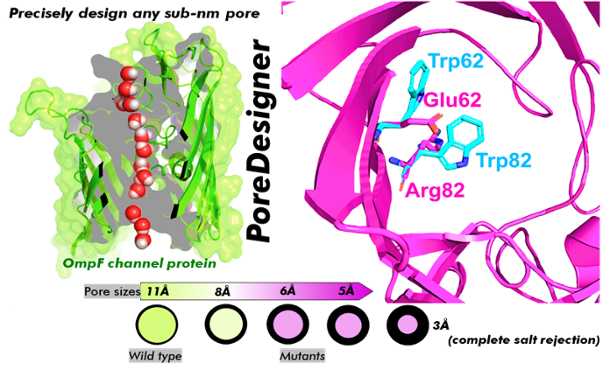
Pore sizes that are more than 10 angstroms (or 1 nm) are an issue for artificial membranes. An angstrom equals one hundred-millionth of a centimeter, which may seem so small that a slight variation would be insignificant. However, to completely reject certain impurities such as salt, the pores need to be of the order of 3 to 4 angstroms.
To achieve this, Chowdhury and the other researchers developed PoreDesigner as a fully automated computational workflow for tuning the pore size of the channel protein OmpF, found in the outer membrane of E. coli bacteria cells.
“The OmpF protein was chosen because it is a more structurally stable molecule than conventionally used aquaporin molecules, so the hypothesis was that if we make changes to the original OmpF molecule to make its pore size smaller it would still retain its structural stability,” Chowdhury said. “The paper validates the hypothesis.”
The pore size of OmpF is 11 angstroms, and the research team reduced the pore size by systematically filling in the pores with hydrophobic, or water-repelling, amino acids.
“From experiments, we found that we could efficiently reject salt with these proteins when put in a membrane assembly,” Chowdhury said.
They also found that it is possible to reduce the pores to specific sizes, ranging from 3 to 10 angstroms, to reject various impurities, creating custom angstrom-scale sieves. The PoreDesigner process also produces water channels that permeate faster than biological water channels. This is due to the hydrophobic inner pore walls of the Ompf protein preventing the water from forming hydrogen bonds with the pore wall, enabling faster transport of water.
There are multiple benefits to the PoreDesigner workflow and the resulting channels. This process saves energy because these water channels are more selective and productive. The PoreDesigner also enables precise pore sizes for bioseparation processes that are extremely difficult, such as separating glucose and fructose, and sucrose from glycine. In addition, because of its effectiveness at filtering out salt, PoreDesigner could potentially enable coastal communities to have a reliable source of water.
Beyond water purification, the researchers are trying to engineer the pores to reject protons while just allowing water to pass through. If successful, this could improve diffusion-weighted MRIs where engineered transport proteins are beginning to be used.
“The higher the flow rate of water through the channels, the better the resolution of the MRI image,” Chowdhury said. “We have shown that our OmpF designs already permeate water at an order of magnitude faster rate than any channel reported, so our designs could be very important for medical imaging.”
Chowdhury added that PoreDesigner bears promise for application in international space stations for filtering urine to get pure water, which would be very important for utilizing and recycling marginal water resources.
Chowdhury’s innovative work on PoreDesigner has received recognition at Penn State and beyond.
"Ratul won the Best Paper award in 2018 for this work from the Department of Chemical Engineering and was recently selected for the North American Membrane Society Student Fellowship,” Manish Kumar, associate professor of chemical engineering. “These accolades are well deserved as Ratul was the driving force for this work and really owned all aspects of the project."
Other researchers from Penn State on this project are Tingwei Ren, doctoral candidate in chemical engineering; Matthew Grisewood, research assistant in chemical engineering; Jeevan Prabhakar, undergraduate researcher in chemical engineering; and Costas Maranas, Donald B. Broughton Professor of Chemical Engineering. Kumar and Maranas co-led the research. Manish Shankla, doctoral candidate at UIUC; Karl Decker, research assistant at UIUC; and Aleksei Aksimentiev, professor of physics at UIUC, also participated in the research.



